Ionization Energy Of Chlorine In Kj/Mol . The electron affinity of chlorine is 349.0 kj mol ‑1. 1 ev / atom = 96.49 kj / mol. With the addition of energy, a lithium ion can be formed from the lithium atom by losing. If an atom possesses more than one electron,. Ionization energy, also called ionization potential, is the. 123 rows molar ionization energies of the elements. for instance, the ionization energy of sodium (alkali metal) is 496kj/mol (1) whereas chlorine's first ionization. Ionisation energies and electron affinity. First ionization energy of chlorine is 12.9676 ev. These tables list values of molar ionization energies, measured in kj⋅mol. look at the equation below: 1st ie = 495.8 kj/mol. typical units for ionization energies are kilojoules/mole (kj/mol) or electron volts (ev): a representation of the atomic spectrum of chlorine.
from www.numerade.com
123 rows molar ionization energies of the elements. The electron affinity of chlorine is 349.0 kj mol ‑1. With the addition of energy, a lithium ion can be formed from the lithium atom by losing. 1 ev / atom = 96.49 kj / mol. look at the equation below: Ionization energy, also called ionization potential, is the. If an atom possesses more than one electron,. 1st ie = 495.8 kj/mol. for instance, the ionization energy of sodium (alkali metal) is 496kj/mol (1) whereas chlorine's first ionization. a representation of the atomic spectrum of chlorine.
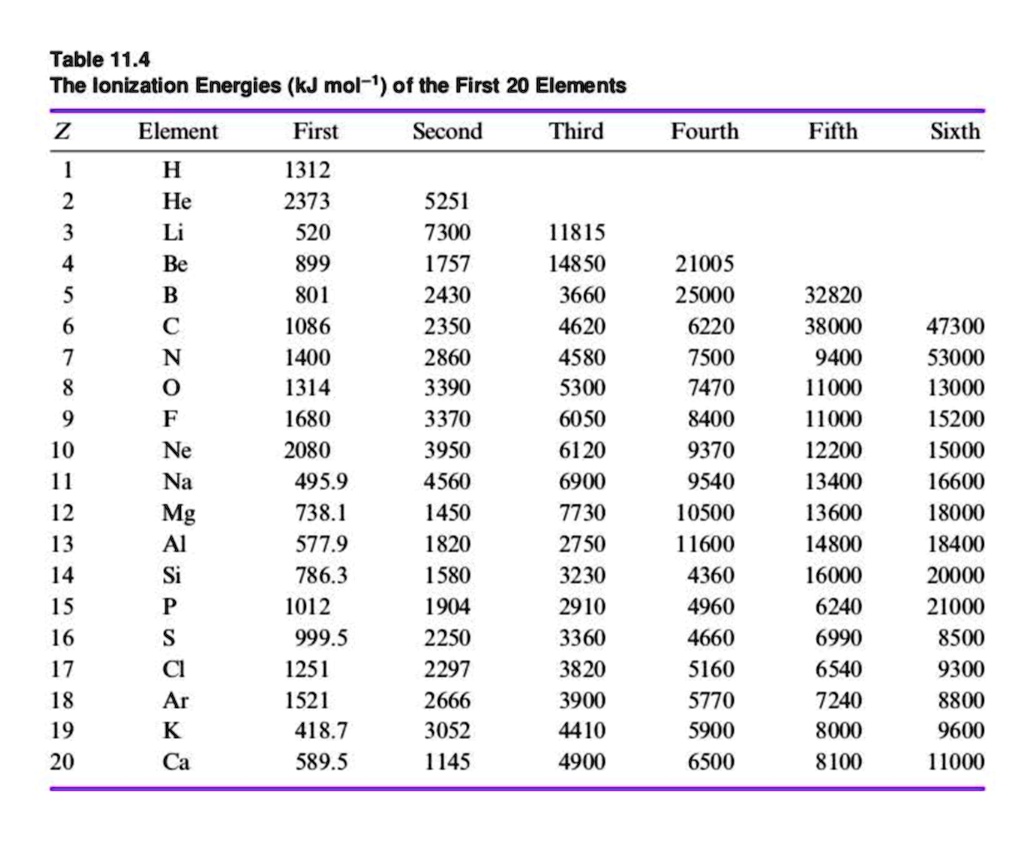
SOLVED Table 11.4 The Ionization Energies (kJ mol1) of the First 20 Elements Element First
Ionization Energy Of Chlorine In Kj/Mol Ionization energy, also called ionization potential, is the. With the addition of energy, a lithium ion can be formed from the lithium atom by losing. The electron affinity of chlorine is 349.0 kj mol ‑1. for instance, the ionization energy of sodium (alkali metal) is 496kj/mol (1) whereas chlorine's first ionization. a representation of the atomic spectrum of chlorine. Ionization energy, also called ionization potential, is the. First ionization energy of chlorine is 12.9676 ev. Ionisation energies and electron affinity. If an atom possesses more than one electron,. These tables list values of molar ionization energies, measured in kj⋅mol. look at the equation below: typical units for ionization energies are kilojoules/mole (kj/mol) or electron volts (ev): 1st ie = 495.8 kj/mol. 123 rows molar ionization energies of the elements. 1 ev / atom = 96.49 kj / mol.
From www.slideserve.com
PPT Periodic Table and Periodic Trends PowerPoint Presentation ID7060128 Ionization Energy Of Chlorine In Kj/Mol a representation of the atomic spectrum of chlorine. The electron affinity of chlorine is 349.0 kj mol ‑1. Ionization energy, also called ionization potential, is the. With the addition of energy, a lithium ion can be formed from the lithium atom by losing. 123 rows molar ionization energies of the elements. 1st ie = 495.8 kj/mol. If an. Ionization Energy Of Chlorine In Kj/Mol.
From www.numerade.com
SOLVED Based on the ionization energies for element "x," predict the formula that would be Ionization Energy Of Chlorine In Kj/Mol typical units for ionization energies are kilojoules/mole (kj/mol) or electron volts (ev): The electron affinity of chlorine is 349.0 kj mol ‑1. a representation of the atomic spectrum of chlorine. These tables list values of molar ionization energies, measured in kj⋅mol. look at the equation below: Ionisation energies and electron affinity. With the addition of energy, a. Ionization Energy Of Chlorine In Kj/Mol.
From cabinet.matttroy.net
Ionization Energy Table Kj Mol Matttroy Ionization Energy Of Chlorine In Kj/Mol typical units for ionization energies are kilojoules/mole (kj/mol) or electron volts (ev): Ionization energy, also called ionization potential, is the. First ionization energy of chlorine is 12.9676 ev. Ionisation energies and electron affinity. 1st ie = 495.8 kj/mol. for instance, the ionization energy of sodium (alkali metal) is 496kj/mol (1) whereas chlorine's first ionization. 123 rows molar. Ionization Energy Of Chlorine In Kj/Mol.
From slideplayer.com
Aim Determining ionization energy and electronegativity of elements ppt download Ionization Energy Of Chlorine In Kj/Mol Ionisation energies and electron affinity. Ionization energy, also called ionization potential, is the. The electron affinity of chlorine is 349.0 kj mol ‑1. for instance, the ionization energy of sodium (alkali metal) is 496kj/mol (1) whereas chlorine's first ionization. 1 ev / atom = 96.49 kj / mol. typical units for ionization energies are kilojoules/mole (kj/mol) or electron. Ionization Energy Of Chlorine In Kj/Mol.
From www.youtube.com
The successive ionization energies (in kJ/mol) for an element are shown below. `{(E_(1),E_(2),E Ionization Energy Of Chlorine In Kj/Mol If an atom possesses more than one electron,. a representation of the atomic spectrum of chlorine. for instance, the ionization energy of sodium (alkali metal) is 496kj/mol (1) whereas chlorine's first ionization. 1st ie = 495.8 kj/mol. Ionisation energies and electron affinity. typical units for ionization energies are kilojoules/mole (kj/mol) or electron volts (ev): These tables list. Ionization Energy Of Chlorine In Kj/Mol.
From www.numerade.com
SOLVEDThe successive ionization energies in kJ/mol of an element P are 740,1500,7000,10500 Ionization Energy Of Chlorine In Kj/Mol a representation of the atomic spectrum of chlorine. 1st ie = 495.8 kj/mol. First ionization energy of chlorine is 12.9676 ev. Ionisation energies and electron affinity. If an atom possesses more than one electron,. The electron affinity of chlorine is 349.0 kj mol ‑1. Ionization energy, also called ionization potential, is the. look at the equation below: . Ionization Energy Of Chlorine In Kj/Mol.
From www.slideserve.com
PPT 103 PowerPoint Presentation, free download ID6775065 Ionization Energy Of Chlorine In Kj/Mol First ionization energy of chlorine is 12.9676 ev. The electron affinity of chlorine is 349.0 kj mol ‑1. look at the equation below: 1 ev / atom = 96.49 kj / mol. typical units for ionization energies are kilojoules/mole (kj/mol) or electron volts (ev): 123 rows molar ionization energies of the elements. for instance, the ionization. Ionization Energy Of Chlorine In Kj/Mol.
From ar.inspiredpencil.com
Successive Ionization Energies Ionization Energy Of Chlorine In Kj/Mol a representation of the atomic spectrum of chlorine. First ionization energy of chlorine is 12.9676 ev. typical units for ionization energies are kilojoules/mole (kj/mol) or electron volts (ev): look at the equation below: Ionization energy, also called ionization potential, is the. Ionisation energies and electron affinity. for instance, the ionization energy of sodium (alkali metal) is. Ionization Energy Of Chlorine In Kj/Mol.
From www.toppr.com
Given, sublimation and ionization energy of Na are 107 kJ/mol and 502 kJ/mol respectively and Ionization Energy Of Chlorine In Kj/Mol If an atom possesses more than one electron,. 1st ie = 495.8 kj/mol. typical units for ionization energies are kilojoules/mole (kj/mol) or electron volts (ev): a representation of the atomic spectrum of chlorine. These tables list values of molar ionization energies, measured in kj⋅mol. look at the equation below: Ionization energy, also called ionization potential, is the.. Ionization Energy Of Chlorine In Kj/Mol.
From www.youtube.com
How do you calculate the ionization energy in kJ/mol of the He+ ion? An H like ion is an ion Ionization Energy Of Chlorine In Kj/Mol 123 rows molar ionization energies of the elements. These tables list values of molar ionization energies, measured in kj⋅mol. for instance, the ionization energy of sodium (alkali metal) is 496kj/mol (1) whereas chlorine's first ionization. If an atom possesses more than one electron,. First ionization energy of chlorine is 12.9676 ev. Ionisation energies and electron affinity. The electron. Ionization Energy Of Chlorine In Kj/Mol.
From www.doubtnut.com
The following table shows the successive molar ionization energ Ionization Energy Of Chlorine In Kj/Mol typical units for ionization energies are kilojoules/mole (kj/mol) or electron volts (ev): Ionisation energies and electron affinity. 1 ev / atom = 96.49 kj / mol. These tables list values of molar ionization energies, measured in kj⋅mol. First ionization energy of chlorine is 12.9676 ev. 1st ie = 495.8 kj/mol. a representation of the atomic spectrum of chlorine.. Ionization Energy Of Chlorine In Kj/Mol.
From www.bartleby.com
Answered Using the data below, calculate the… bartleby Ionization Energy Of Chlorine In Kj/Mol a representation of the atomic spectrum of chlorine. look at the equation below: 1 ev / atom = 96.49 kj / mol. 123 rows molar ionization energies of the elements. for instance, the ionization energy of sodium (alkali metal) is 496kj/mol (1) whereas chlorine's first ionization. First ionization energy of chlorine is 12.9676 ev. Ionization energy,. Ionization Energy Of Chlorine In Kj/Mol.
From www.numerade.com
SOLVED Anhydrous AlCl3 is a covalent compound. From the data given below, predict whether it Ionization Energy Of Chlorine In Kj/Mol With the addition of energy, a lithium ion can be formed from the lithium atom by losing. First ionization energy of chlorine is 12.9676 ev. 1st ie = 495.8 kj/mol. 123 rows molar ionization energies of the elements. These tables list values of molar ionization energies, measured in kj⋅mol. Ionization energy, also called ionization potential, is the. look. Ionization Energy Of Chlorine In Kj/Mol.
From askfilo.com
Example 16 A single electron system has ionization energy 11180 kJ mol−1.. Ionization Energy Of Chlorine In Kj/Mol for instance, the ionization energy of sodium (alkali metal) is 496kj/mol (1) whereas chlorine's first ionization. Ionization energy, also called ionization potential, is the. typical units for ionization energies are kilojoules/mole (kj/mol) or electron volts (ev): 1 ev / atom = 96.49 kj / mol. a representation of the atomic spectrum of chlorine. With the addition of. Ionization Energy Of Chlorine In Kj/Mol.
From byjus.com
29. The first,second and third ionization energies are 578,1187and 2745 kj/mol respectively Ionization Energy Of Chlorine In Kj/Mol look at the equation below: Ionisation energies and electron affinity. These tables list values of molar ionization energies, measured in kj⋅mol. The electron affinity of chlorine is 349.0 kj mol ‑1. a representation of the atomic spectrum of chlorine. Ionization energy, also called ionization potential, is the. If an atom possesses more than one electron,. 123 rows. Ionization Energy Of Chlorine In Kj/Mol.
From www.doubtnut.com
The first ΔiH1 and the second ΔiH ionization enthalpies in kJ mol Ionization Energy Of Chlorine In Kj/Mol The electron affinity of chlorine is 349.0 kj mol ‑1. If an atom possesses more than one electron,. typical units for ionization energies are kilojoules/mole (kj/mol) or electron volts (ev): for instance, the ionization energy of sodium (alkali metal) is 496kj/mol (1) whereas chlorine's first ionization. Ionization energy, also called ionization potential, is the. 1st ie = 495.8. Ionization Energy Of Chlorine In Kj/Mol.
From www.doubtnut.com
The successive ionization energies (in kJ/mol) for an element are shown below. {(E(1),E(2),E(3 Ionization Energy Of Chlorine In Kj/Mol First ionization energy of chlorine is 12.9676 ev. With the addition of energy, a lithium ion can be formed from the lithium atom by losing. a representation of the atomic spectrum of chlorine. look at the equation below: typical units for ionization energies are kilojoules/mole (kj/mol) or electron volts (ev): 1st ie = 495.8 kj/mol. The electron. Ionization Energy Of Chlorine In Kj/Mol.
From www.slideserve.com
PPT Chapter 5 The Periodic Law PowerPoint Presentation, free download ID5937138 Ionization Energy Of Chlorine In Kj/Mol typical units for ionization energies are kilojoules/mole (kj/mol) or electron volts (ev): 123 rows molar ionization energies of the elements. a representation of the atomic spectrum of chlorine. These tables list values of molar ionization energies, measured in kj⋅mol. 1st ie = 495.8 kj/mol. With the addition of energy, a lithium ion can be formed from the. Ionization Energy Of Chlorine In Kj/Mol.